Molecular mechanism and function of neuronal autophagy
VOLKER HAUCKE (FMP, Berlin)
FAN LIU (FMP, Berlin)


Neurons depend on spatially organized and tightly regulated trafficking pathways to maintain proteostasis across their highly polarized architecture. In the first funding period, we identified Golgi satellites (GS) which are small, trans-Golgi-like subcompartments to be present not only in dendrites but also in axons, including presynaptic boutons. We found that myosin VI mediates their activity-dependent localization to synapses, and that impairing this targeting could disrupts local degradative capacity, reflected by increased autophagosome accumulation. In search for alternative routes which would allow neurons to remove degraded components without a need to be transported to the soma, we obtained evidence for secretion of mitochondrial components, suggesting that secretory mitophagy may act as a constitutive clearance pathway in neurons. Building on these findings, the proposed project will investigate two central objectives in the second funding period. First, we will define the role of myosin VI in coordinating the maturation and dynamics of GS, lysosomes, and autophagosomes at presynaptic boutons. We will test whether GS serve as a local source of degradative enzymes, facilitating autolysosomal maturation through spatially organized organelle interactions. Second, we will examine how synaptic activity influences the ability of presynaptic boutons to engage in secretion of degradative cargo, focusing on the molecular mechanisms and checkpoints that govern the routing of organelles between degradation and exocytosis. This work will advance our understanding of spatial proteostasis in neurons and uncover new principles of local degradation and secretion that are essential for maintaining synaptic integrity
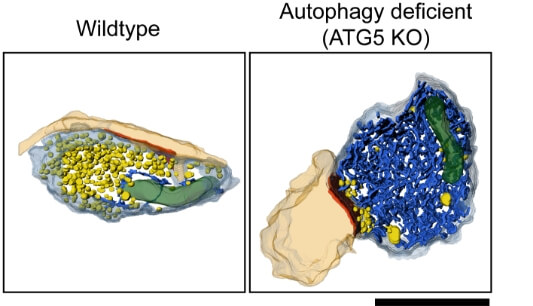
3D TEM tomography reconstructions of synaptic boutons showing postsynaptic densities (orange), ER tubules (blue), synaptic vesicles (yellow) and mitochondria (green). Examples are showing a wildtype bouton and an autophagy-deficient (ATG5-KO) bouton with severe ER volume increases. Scale bar, 1 μm.
References:
Anja Konietzny, Yuhao Han, Yannes Popp, Bas van Bommel, Aditi Sharma, Philippe Delagrange, Nicolas Arbez, Marie-Jo Moutin, Leticia Peris, Marina Mikhaylova; Efficient axonal transport of endolysosomes relies on the balanced ratio of microtubule tyrosination and detyrosination. J Cell Sci 15 April 2024; 137 (8): jcs261737. doi: https://doi.org/10.1242/jcs.261737
Hertrich, N., Han, Y., Doil, N., Dimitra Ranti, Britta Eickholt, Anja Konietzny, Marina Mikhaylova;. Myosin VI controls localization of golgi satellites at active presynaptic boutons. Cell. Mol. Life Sci. 82, 357 (2025). https://doi.org/10.1007/s00018-025-05896-2
Subkhangulova, A., Mikhaylova, M. The Golgi apparatus: adaptations to neuronal shape and functions. EMBO J 45, 358–373 (2026). https://doi.org/10.1038/s44318-025-00658-z
