Maintaining presynaptic function in dopaminergic synapses
CHRISTIAN ROSENMUND (Charité Universitätsmedizin, Berlin)
CRAIG C. GARNER (Deutsche Zentrum für Neurodegenerative Erkrangungen)
ECKART D. GUNDELFINGER (Leibniz Institute for Neurobiology (LIN), Magdeburg)



Midbrain dopaminergic neurons have important functions in the brain, including modulation of motor functions, motivation, decision-making and reward. Loss of these neurons is associated with neurodegenerative disorders, such as Parkinson’s disease and others. The reasons for the high susceptibility of dopaminergic neurons to degeneration are still largely enigmatic. We hypothesize that early events of dysfunction take place at dopamine release sites and originate from disturbed presynaptic proteostasis and membrane trafficking pathways involving different clearance systems. To test this hypothesis, we use genetic, electrophysiological and cellular imaging tools and techniques that we have successfully applied to study clearance mechanisms at glutamatergic synapses. With those in hand, we will investigate membrane trafficking processes underlying proteostasis at dopaminergic boutons and their role in maintaining presynaptic health. To this end, we aim…
– to identify and characterize the local regulators of presynaptic proteostasis in dopaminergic boutons;
– to define how changes in proteostasis affect (co-)release properties and ultrastructure near dopaminergic release sites; and
– to study pathological mechanisms in dopaminergic (DA) boutons and test the hypothesis that conditions that enhance local surveillance and clearance of presynaptic proteins can be neuroprotective.
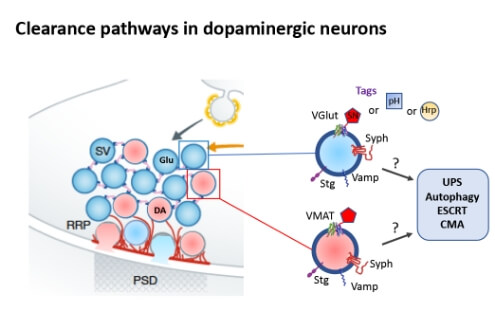
Fig. 1 Dopaminergic boutons release glutamate and dopamine, putatively from two different types of vesicles. To identify turnover pathways of these vesicles and their integral proteins we will tag specific vesicular proteins with supernova (SN), pHluorin (pH) or horseradish peroxidase (Hrp) and study the clearance systems involved in their degradation.
References:
Hoffmann S, Orlando M, Andrzejak E, Bruns C, Trimbuch T, Rosenmund C, Garner CC, Ackermann, F. (2019). Light-Activated ROS Production Induces Synaptic Autophagy. J Neurosci 39, 2163-2183.
Hoffmann-Conaway S, Brockmann MM, Schneider K, Annamneedi A, Rahman KA, Bruns C, Textoris-Taube K, Trimbuch T, Smalla KH, Rosenmund C, Gundelfinger ED, Garner CC, Montenegro-Venegas C. (2020). Parkin contributes to synaptic vesicle autophagy in Bassoon-deficient mice. eLife 9, e56590.
Okerlund N, Schneider K. Leal-Ortiz S, Montenegro-Venegas C, Kim SA., Garner L, Waites C, Gundelfinger ED, Reimer RJ, Garner CC (2017). Bassoon Controls Presynaptic Autophagy through Atg5. Neuron 93, 897-913 e897
Zimmermann J, Herman MA, Rosenmund C (2015). Co-release of glutamate and GABA from single vesicles in GABAergic neurons exogenously expressing VGLUT3. Front Synaptic Neurosci. doi: 10.3389/fnsyn.2015.00016.
Waites, C.L., Leal-Ortiz, S.A., Okerlund, N., Dalke, H., Fejtova, A., Altrock, W.D., Gundelfinger, E.D., and Garner, C.C. (2013). Bassoon and Piccolo maintain synapse integrity by regulating protein ubiquitination and degradation. EMBO J 32, 954-969.


