Interdependencies of autophagy, protein synthesis, aging, activity and synaptic viability
DANIELA C. DIETERICH (Otto-von-Guericke University, Magdeburg)
NOAM E. ZIV (Technion Faculty of Medicine and Network Biology Labs, Haifa)


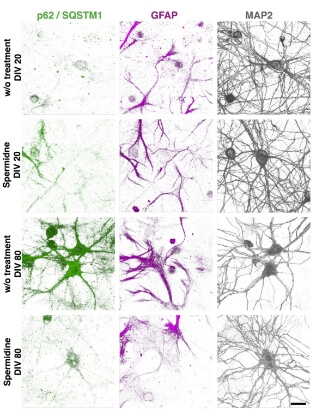
Synapses in the brain can persist for months and even years. Their proteinaceous components, however, become dysfunctional after much shorter periods, and thus must be continuously removed and degraded. Autophagy is one such removal pathway, mainly involved in clearance of protein complexes and aggregates. Significant evidence suggests that aging is associated with impaired protein clearance, and that manipulations that augment autophagy increase life-span and rejuvenate multiple physiological processes including several pertaining to synaptic and cognitive functions. Catabolic and anabolic processes are often coupled, and thus manipulations that enhance autophagy are likely to affect other aspects of protein metabolism. Our overall goal in this project is thus to gain a broader view of the effects exerted by such manipulations, using them to expose interdependencies among autophagy, protein synthesis, aging, activity, and synaptic viability. To that end we will examine how manipulations of autophagy affect (synaptic) protein synthesis and degradation in standard and aged neuronal cultures, in mice of different ages, and in mice raised in enriched environments. Long-term imaging will be used to examine how these manipulations affect autophagic flux, neuronal viability, synaptic persistence, tenacity and function, as well as resilience to stressors. Ultimately, we hope to use the obtained data to identify autophagy-associated targets for manipulations aimed at improving life-long neuronal and synaptic viability and test a subset of these within the consortium.
References:
Cohen LD, Ziv NE. (2019) Neuronal and synaptic protein lifetimes. Curr Opin Neurobiol. 57:9-16
Ziv NE. (2018) Maintaining the active zone: Demand, supply and disposal of core active zone proteins. Neurosci Res. 127:70-77.
Hakim V, Cohen LD, Zuchman R, Ziv T, Ziv NE. (2016) The effects of proteasomal inhibition on synaptic proteostasis. EMBO J.35:2238-2262.
Alvarez-Castelao B, Schanzenbacher CT, Hanus C, Glock G, Tom Dieck S, Dorrbaum AR, Bartnik I, Nassim-Assir B, Ciirdaeva E, Mueller A, Dieterich DC, Tirrell DA, Langer JD, Schuman EM. (2017) Cell-type-specific metabolic labeling of nascent proteomes in vivo. Nat Biotechnol doi: 10.1038/nbt.4016.
Dieterich DC, Kreutz MR (2016) Proteomics of the synapse – A quantitative approach to neuronal plasticity. Mol Cell Proteomics 15:368–381.
Dieterich DC, Hodas JJ, Gouzer G, Shadrin IY, Ngo JT, Triller A, Tirrell DA, Schuman EM (2010) In situ visualization and dynamics of newly synthesized proteins in rat hippocampal neurons. Nature Neurosci 13, 897-905

